주제 3 급성 뇌졸중 발병시간 추정

Scientific overview (연구 배경 및 중요성)
Determination of symptom onset time for acute ischemic stroke is crucial because of the
treatment options highly depend onthe time window. Previously intravenous recombinant
tissue plasminogen activator (rt-PA) is available up to 4.5 hours from symptom onset.
Therefore, at 2018 HeLP challenge was opened for classifying strokes into those
within 4.5h and those over 4.5h from stroke onset. In consideration of intra-arterial thrombectomy,
recent HeLP challenge at 2021 was opened for classifying strokes into those within 6h and those over
6h from stroke onset. However, there have been limitations in its extended application by classification-based
AI developments.
It is still difficult to pinpoint the onset of stroke in those with wake-up stroke or unwitnessed daytime stroke.
For these patients, tissue clock instead of chronological clock was raised as an alternative.
Multiparametric magnetic resonance imaging (MRI), which uses multiple sequences sensitive to
different aspects of tissue pathophysiology in acute stroke, has been used to estimate the stroke
onset time when it is unknown. For example, alterations of water diffusion can be detected by a reduced
apparent diffusion coefficient (ADC) on diffusion-weighted imaging (DWI) within minutes of stroke,
whereas a net increase of water content can be detected as in increased T2 signal within 1 to 4 hours
from ischemia onset. Based on these distinctive characteristics, the mismatch in the visibility of an
acute ischemic lesion between DWI and fluid-attenuated inversion recovery (FLAIR) has been most commonly studied.
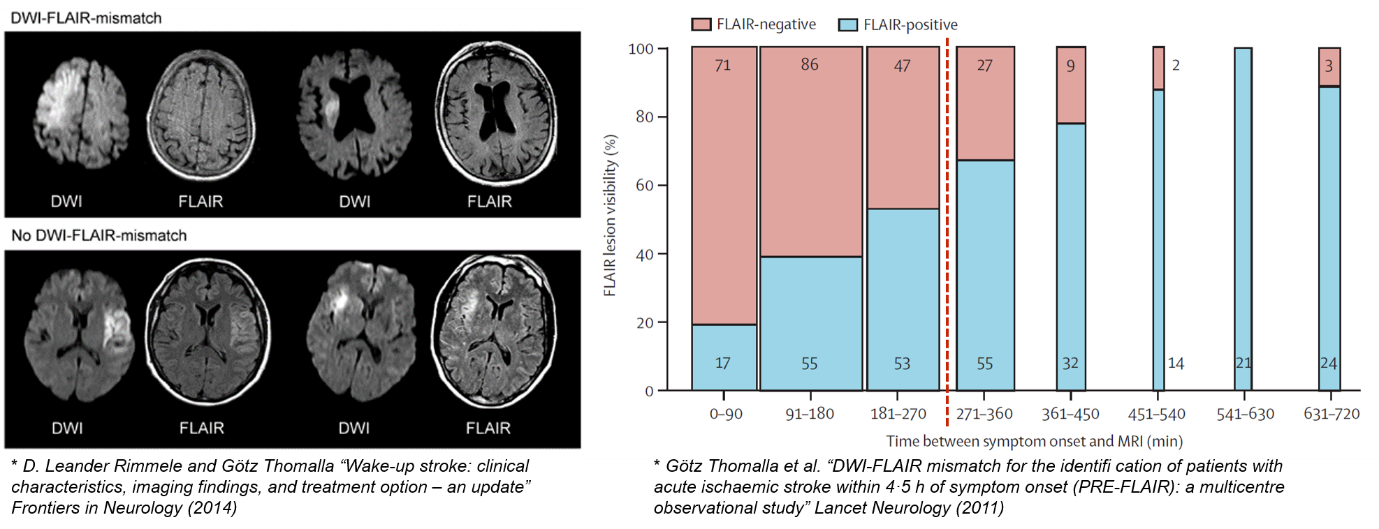
Given the promise of machine learning and deep learning to aid in identifying the tissue age of acute stroke, specifically, exact hours from onset, we propose the HeLP Challenge 2023 – Stroke Onset Time Prediction, an open crowd sourced algorithmic analysis challenge to better predict time since symptom onset by acute ischemic stroke. An improved diagnostic tool may be feasible and useful in discriminating patients who would be candidates for re-perfusion therapy (e.g., rt-PA, intra-arterial thrombectomy) among those with unclear onset stroke or those in the expanded time window
Challenge questions (문제 정의)
Develop a predictive method to accurately interpret the hours of MRI scans from symptom onset of stroke using multi-modal MRIs (i.e., DWI and FLAIR)
Data description (데이터 설명 – 데이터 셋 구성, 형식, 특징)
Data collection from multi-center (4 institutes in South Korea) will be provided as the training and validation data for this year’s HeLP challenge - Stroke Onset Time Prediction.
- (1) Data Set : 221 of acute stroke patients (155 and 66 for Training and Validation, respectively)
- (2) Clinical Data and Label : Hours of MRI scans from symptom onset of stroke (training data only)
- (3) Image Data : Diffusion-weighted MRI (DWI), fluid attenuated inversion recovery (FLAIR) and infarct lesion mask
MRI scans were performed using 3 types of 1.5-T clinical whole body scanners from different vendors (Avanto, Siemens; Signa, GE Medical Systems; Achieva, Philips Medical Systems) with a standard head coil. For each patient, an acute stroke MRI protocol including DWI and FLAIR sequences was used in each center. ADC maps were automatically created from DWI scans using the built-in software. Therefore, the imaging parameters are different depending on institutes and vendors. Representative imaging parameters for each type of scanner are summarized in the Table below.
| GE Medical System | Philips Medical System | Siemens | ||
|---|---|---|---|---|
| DWI | b-value (s/mm2) | 0 and 1,000 | ||
| TR (ms) / TE (ms) | 7,000 / 86 | 3,000 / 56 | 3,000 / 86 | |
| FOV (mm x mm) | 250 x 250 | |||
| Matrix size | 256 x 256 | 256 x 256 | 384 x 384 | |
| Number of slices | 20 | |||
| Slice thickness (mm) / gap (mm) | 5.0 / 2.0 | |||
| FLAIR | TR (ms) / TE (ms) / TI (ms) | 10,000 / 97.5 / 2,200 | 11,000 / 125 / 2,800 | 9,000 / 100 / 2,500 |
| FOV (mm x mm) | 250 x 250 | 230 x 230 | 190 x 210 | |
| Matrix size | 256 x 256 | 512 x 512 | 232 x 256 | |
| Number of slices | 20 | |||
| Slice thickness (mm) / gap (mm) | 5.0 / 2.0 | |||
To help to focus on the DWI-FLAIR mismatch within infarct lesions, the segmented mask of infarct lesions were also provided. Infarct regions were manually segmented on the DWI maps by a biomedical engineer. And then, one stroke neurologists assessed the segmentation results and manually corrected the wrong segmented mask.
The brain MRI images and infarct lesion mask are provided in compressed NifTi format (.nii.gz). For each patient, the provided FLAIR image was co-registered to its native space (i.e., DWI acquisition space). Registration was applied using ANTs Toolbox: from its native space to Montreal Neurological Institute (MNI) space with a resolution of 2 mm × 2 mm × 2 mm (91 x 109 x 91 voxel in x-, y-, and z-axis).
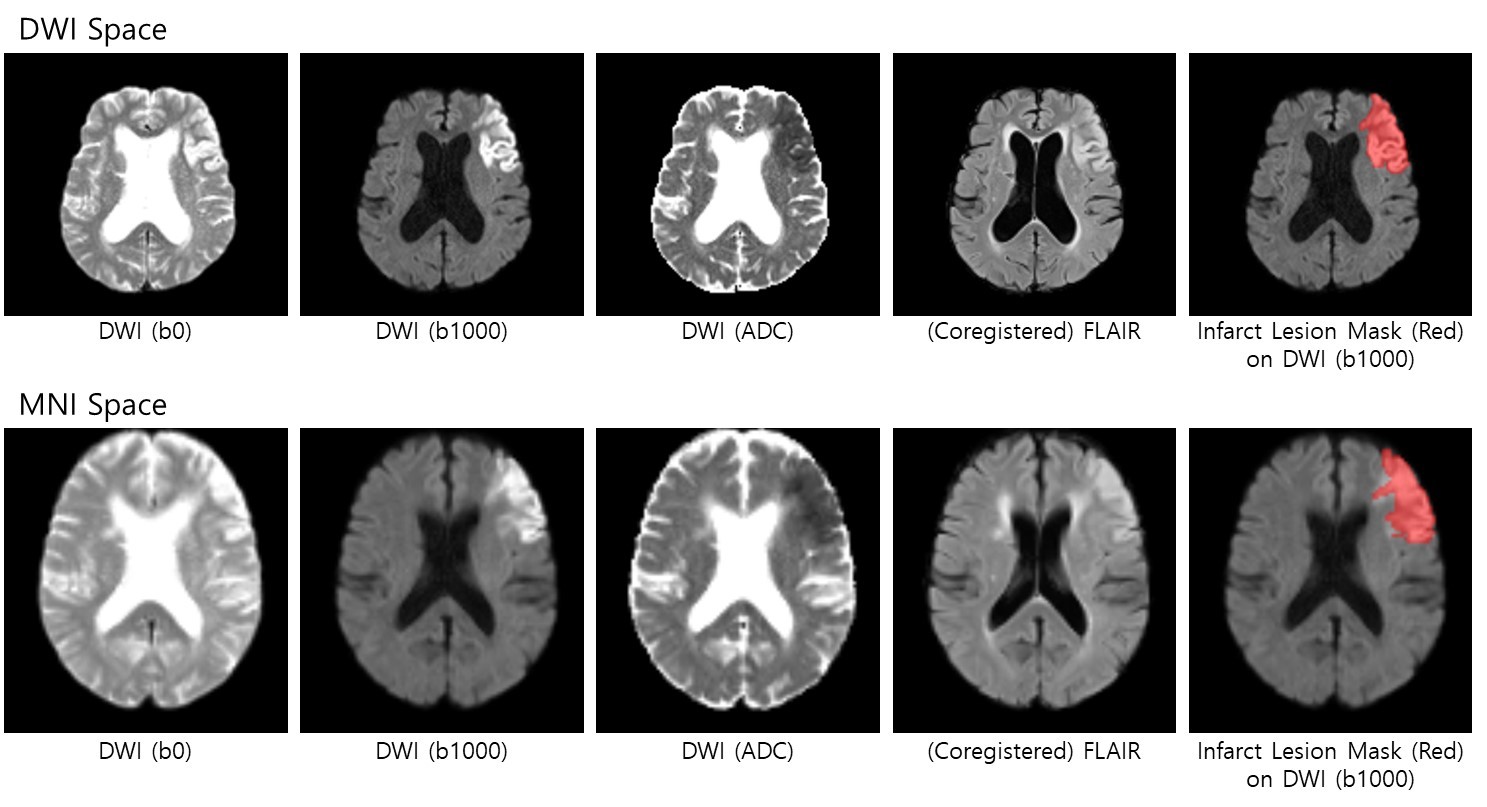
Each patient have two folders based on preprocessing (i.e., DWI space or MNI space). Each folder contains 5 files of images as followed:
[DWI_Space] Folder (5 files): Brain Extraction was applied. FLAIR was co-registered to DWI.- (1) b0_brain.nii.gz
- (2) b1000_brain.nii.gz
- (3) adc_brain.nii.gz
- (4) lesion1_brain.nii.gz (manually annotated lesion mask)
- (5) flair_in_b0_brain.nii.gz (co-registered to DWI b0 image)
- (1) b0_brain_MNI_iso2mm.nii.gz
- (2) b1000_brain_MNI_iso2mm.nii.gz
- (3) adc_brain_MNI_iso2mm.nii.gz
- (4) lesion1_brain_MNI_iso2mm.nii.gz
- (5) flair_in_b0_brain_MNI_iso2mm.nii.gz
Evaluation matrix (정량적 평가 방법)
(1) Requirements for participants in submission (excel)
- Submit the predicted hours of MRI scans from symptom onset of stroke by the developed methods (floating point number)
(2) Evaluation
- Root-mean-squared-error between submitted and labeled scores in validation data




